The field of medical robotics represents one of the most dynamic and fastest-growing intersections of modern technology and healthcare. Driven by a global surge in demand for high-precision, minimally invasive, and assistive care, this domain is rapidly evolving, drawing together experts from disciplines as diverse as medicine, robotics, computer science, artificial intelligence (AI), biomedical engineering, and materials science. The proliferation of new research, innovative products, and novel applications creates a vibrant but complex landscape. This article aims to synthesize the current state of medical robotics from a unified, interdisciplinary viewpoint. By re-examining its conceptual foundations, classifying its diverse applications, and analyzing its core technological principles and challenges, we seek to provide a coherent framework that fosters mutual understanding and collaboration across the many fields contributing to the advancement of the medical robot.
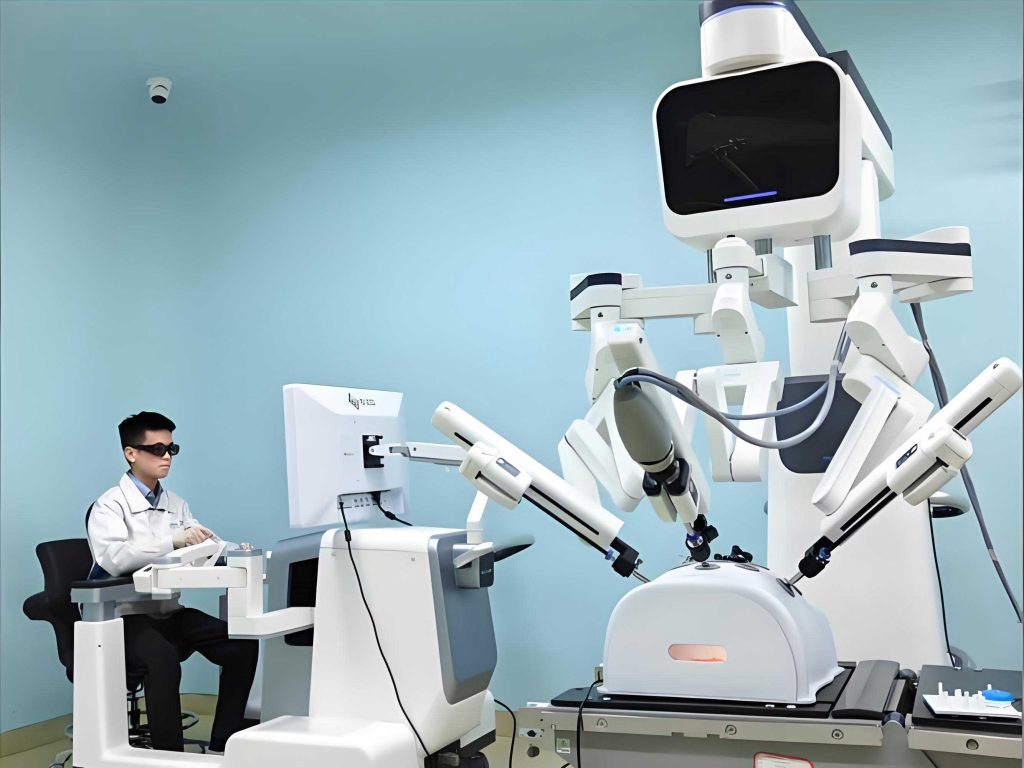
Traditionally, definitions of robotics, including medical robot systems, have been anchored in hardware—the mechanical arm, the mobile base, the articulated tool. This perspective, inherited from industrial automation, is increasingly inadequate. A modern medical robot is not merely a device; it is a sophisticated cyber-physical system where critical functions are distributed between human operators (surgeons, therapists, clinicians) and technological components (sensors, algorithms, actuators). In many essential roles, particularly high-level decision-making and situational judgment, the human remains irreplaceable and central to the system’s operation. Therefore, this article proposes a paradigm shift: to define a medical robot as a human-machine hybrid system architected around a core set of five integrated functions. This functional framework offers a more accurate and comprehensive lens through which to understand, design, and evaluate all categories of medical robot applications.
Classification of Medical Robots
The spectrum of medical robot applications is broad and can be logically categorized based on primary purpose. While boundaries between categories can sometimes blur, a clear classification aids in understanding the technological focus and clinical requirements of each type. The primary classes are Surgical Robots, Medical Assistance Robots, Rehabilitation Robots, and Hospital Service Robots.
| Primary Class | Sub-class (Examples) | Typical Applications & Notes |
|---|---|---|
| Surgical Robots | Laparoscopic/Robotic-Assisted Minimally Invasive Surgery (MIS) Robots | Prostatectomy, cholecystectomy, cardiac, gynecological, and general soft-tissue surgery. Characterized by master-slave teleoperation. |
| Orthopedic Surgery Robots | Total knee/hip arthroplasty, spinal pedicle screw placement, osteotomy. Focus on bone machining and precise implant positioning. | |
| Neurosurgical Robots | Biopsy, deep brain stimulation (DBS) electrode placement, stereo-EEG. Require extreme precision and stability. | |
| Vascular Interventional Robots | Percutaneous coronary intervention (PCI/stenting), cardiac ablation. Remote navigation of catheters and guidewires under fluoroscopy. | |
| Medical Assistance Robots | Diagnostic & Biopsy Robots | Robotically-steered ultrasound probes, automated breast biopsy systems, capsule endoscopy locomotion control. |
| Logistics & Pharmacy Robots | Automated intravenous (IV) drug compounding, blood drawing robots, automated pharmacy dispensing systems. | |
| Rehabilitation Robots | Rehabilitation Therapy Robots | Upper/lower limb exoskeletons and end-effector devices for post-stroke motor relearning, gait training. |
| Functional Assistive Robots | Intelligent prosthetic limbs, powered wheelchairs, robotic feeding arms, socially assistive robots for cognitive therapy (e.g., autism). | |
| Hospital Service Robots | Transport & Logistics Robots | Autonomous mobile robots (AMRs) for delivery of medications, lab samples, linens, and sterile supplies. |
| Disinfection & Sanitation Robots | Ultraviolet (UV-C) light or hydrogen peroxide vapor disinfection robots. |
The Functional Architecture of a Medical Robot System
Underlying the diverse forms in Table 1 is a common functional architecture. Every medical robot, from a neurosurgical platform to a logistics AMR, can be described as a system integrating five core functions: Perception, Data, Planning, Execution, and Control. The implementation and balance of automation versus human input within each function vary significantly across applications, but their interrelationships define the system’s fundamental workflow.
1. Perception: This function is responsible for acquiring all relevant information about the patient, the environment, and the robot itself. It forms the sensory foundation of the system.
$$ \text{Sensory Data} = \{ I_{\text{pre-op}}(CT, MRI, …), I_{\text{intra-op}}(Video, Force, EM, …), I_{\text{env}}(Lidar, RGB-D, …) \} $$
Inputs come from medical imaging (pre-operative CT/MRI, intra-operative ultrasound/fluoroscopy), robot-mounted sensors (optical trackers, force/torque sensors, endoscopic cameras), and environmental sensors (LiDAR, depth cameras for navigation). The output feeds directly into the Control function for real-time reaction and into the Data function for processing and modeling.
2. Data: This function manages, processes, and interprets the information from Perception. Its key role is to create and maintain actionable models.
$$ \text{Patient Model } \mathcal{M}(t) = \mathcal{F}(\text{Sensory Data}, \text{Prior Knowledge}) $$
It involves tasks like medical image segmentation, 3D anatomical model reconstruction, sensor fusion for real-time tracking, and integration with electronic health records (EHR). The Data function outputs structured information (e.g., a registered 3D model of a vertebra) to both the Planning and Control functions.
3. Planning: This function formulates the sequence of actions needed to achieve a medical objective. Currently, it is predominantly a human-driven function with computational support.
$$ \text{Plan } \mathcal{P} = \arg\max_{\mathcal{A}} U(\mathcal{A} | \mathcal{M}, \mathcal{K}) $$
where $\mathcal{A}$ is a sequence of actions, $U$ is a utility function (e.g., maximizing safety, minimizing tissue damage), $\mathcal{M}$ is the patient model, and $\mathcal{K}$ is clinical knowledge. In surgery, this is the pre-operative planning of screw trajectories or resection margins. In rehabilitation, it is the design of a therapy exercise regimen. The plan $\mathcal{P}$ is primarily sent to the Control function to guide the human operator, or directly to the Execution function for autonomous tasks.
4. Execution: This function carries out physical or informational actions. It comprises the electromechanical hardware (actuators, manipulators, mobile bases) and software agents that produce the intended effect.
$$ \text{Action } a(t) = G(\text{Control Signal } u(t), \text{ or Plan } \mathcal{P}) $$
For example, a surgical robot’s manipulator makes an incision, a rehabilitation exoskeleton moves a patient’s limb, or a logistics robot drives to a specified location. It receives commands either from the Control function (human-in-the-loop) or directly from the Planning function (autonomous operation).
5. Control: This is the central function that closes the loop between perception and action, enabling task performance. In most medical robot systems, it is a shared responsibility between human and machine.
$$ u(t) = C_{\text{human}} + C_{\text{auto}}(e(t), \mathcal{M}, \mathcal{P}) $$
Here, $u(t)$ is the control signal sent to the Execution function, $C_{\text{human}}$ is the input from the surgeon’s console or therapist’s interface, and $C_{\text{auto}}$ is the automated control component (e.g., tremor filtering, virtual fixture enforcement, path following). The Control function uses real-time feedback from Perception and guidance from the Plan to generate these signals. It embodies the critical decision-making layer, which remains largely human-supervised.
| Function | Surgical Robot (e.g., Spinal) | Rehabilitation Robot (e.g., Exoskeleton) | Hospital Service Robot (e.g., AMR) |
|---|---|---|---|
| Perception | Pre-op CT, intra-op optical/EM tracker, endoscopic video. | IMU sensors, joint encoders, force sensors, sometimes EMG/EEG. | LiDAR, cameras, ultrasonic sensors for mapping & obstacle detection. |
| Data | 3D vertebral model reconstruction, registration to tracker space. | Biomechanical model of limb, estimation of patient’s movement intent. | Dynamic map of hospital floor, location of targets (rooms, stations). |
| Planning | Surgeon plans optimal pedicle screw trajectory on 3D model. | Central system assigns delivery tasks; robot plans optimal path. | |
| Execution | Robotic arm precisely guides drill along planned trajectory. | Actuators move exoskeleton joints to assist patient’s gait. | Wheel motors navigate the robot along the planned path. |
| Control | Surgeon oversees and initiates steps; robot provides steady guidance and constraints (“virtual fixtures”). | Robot provides adaptive assistance based on patient’s real-time performance (assist-as-needed control). | Fully autonomous navigation control, with remote human override capability. |
Key Technological Challenges and Frontiers
The evolution of the medical robot is propelled by efforts to address persistent technical challenges. Solving these is crucial for enhancing safety, efficacy, accessibility, and autonomy.
1. Effective Haptic Feedback (Force & Tactile Sensing): The lack of authentic force feedback in master-slave surgical systems remains a significant limitation. Surgeons rely on visual cues instead of the natural haptic sensations of tissue compliance, vessel pulsation, or suture tension. This can lead to increased tissue forces and potential injury. Research focuses on integrated force/torque sensors, model-based force estimation algorithms, and novel cutaneous tactile displays that provide cues without restricting instrument motion. The challenge is multifold: miniaturization for in-tool sensing, stability of estimation algorithms, and creating an intuitive feedback modality. The ideal system would transmit forces $F_{\text{contact}}$ such that:
$$ F_{\text{master}} = K \cdot F_{\text{contact}} + B $$
where $F_{\text{master}}$ is the force rendered to the surgeon, $K$ is a scalable gain, and $B$ represents any necessary bias or filtering for stability and clarity.
2. Dynamic Target Tracking and Motion Compensation: Physiological motion (respiration, cardiac cycle) and tissue deformation during intervention cause target anatomy to shift. A medical robot must track this motion in real-time. While optical tracking with fiducial markers is common, it suffers from line-of-sight constraints. Alternative approaches include:
- Model-Image Fusion: Continuously registering pre-operative models ( $\mathcal{M}$ ) to intra-operative images (e.g., ultrasound, fluoroscopy).
- Deformable Registration: Using algorithms to update $\mathcal{M}(t)$ in real-time based on sparse sensor data.
- Embedded Sensors: Exploring miniature EM trackers or fiducials detectable in multiple imaging modalities.
The goal is to maintain a real-time estimate of the target pose $X_{\text{target}}(t)$ to guide the robot accordingly.
3. Enhanced Decision Support and Context-Aware Autonomy: Moving beyond mere tool guidance, the next frontier is providing intelligent cognitive assistance. This involves AI and machine learning for:
- Surgical Phase Recognition: Understanding the current step of a procedure from video and robot kinematics.
- Anatomical Landmarking & Segmentation: Automatically identifying critical structures in real-time imaging.
- Predictive Analytics: Warning of potential complications (e.g., vessel proximity) based on the surgical plan and real-time data.
- Automated Sub-task Execution: Enabling supervised autonomy for repetitive, well-defined tasks like suturing or debridement.
The challenge lies in ensuring these systems are robust, interpretable, and safe in the face of immense anatomical and pathological variability.
4. Defining and Achieving Appropriate Autonomy Levels: Full autonomy in complex medical interventions is neither feasible nor desirable in the foreseeable future. The focus is on improving semi-autonomous collaboration. A useful taxonomy defines levels from L1 (Robot Assistance) to L5 (Full Autonomy). Most advanced medical robot systems today operate at L2 (Task Autonomy, e.g., autonomous drilling of a pre-planned bore) or L3 (Conditional Autonomy). The key research direction is developing shared control paradigms where the robot’s autonomy $C_{\text{auto}}$ dynamically adapts to the context, the operator’s skill, and task criticality, always under a “human-on-the-loop” supervisory model. The objective is to reduce outcome variability dependent on operator skill, not to remove the operator.
| Technology | Synergy with Medical Robotics | Potential Impact |
|---|---|---|
| 5G & Edge Computing | Enables stable, low-latency teleoperation for remote surgery and telerehabilitation, breaking geographical barriers to expert care. | Makes telesurgery and remote procedural guidance clinically viable. |
| Soft Robotics | Provides inherently safe, compliant actuators for physical human-robot interaction (pHRI). | Revolutionizes rehabilitation exoskeletons, assistive feeding arms, and novel minimally invasive surgical tools that can navigate delicate anatomy. |
| Brain-Computer Interface (BCI) | Directly decodes patient’s movement intent or cognitive state from neural signals. | Creates more natural control paradigms for neuroprosthetics and rehabilitation robots, enabling patient-driven therapy. |
| Micro/Nanorobotics | Develops robots at cellular scales for intervention within the body. | Opens new frontiers in targeted drug delivery, in-vivo sensing, and microsurgery (e.g., clearing clogged arteries). |
| Advanced AI & Computer Vision | Provides the “perceptual intelligence” for scene understanding, instrument tracking, and tissue characterization. | Essential for the decision support and context-aware autonomy discussed above, making robots more perceptive and responsive. |
Conclusion
The journey of the medical robot from a conceptual tool to a central pillar of modern healthcare is accelerating. By adopting a functional architecture perspective—viewing every system as an integration of Perception, Data, Planning, Execution, and Control—we gain a powerful, unified framework to comprehend its diverse manifestations, from the operating room to the rehabilitation clinic and hospital corridor. This framework underscores the fundamental nature of the medical robot as a human-machine partnership, where technology amplifies human skill, extends human reach, and compensates for human limitations, but does not replace the clinician’s judgment.
The path forward is paved with significant interdisciplinary challenges: restoring the sense of touch, tracking living anatomy, providing intelligent assistance, and defining safe, effective levels of autonomy. Progress will not come from robotics alone, but from deep convergence with AI, material science, biomechanics, and neuroscience. As these challenges are met, the medical robot will evolve into an even more seamless, intuitive, and capable partner, ultimately making high-quality, personalized medical and assistive care more precise, accessible, and consistent across the globe. The future of the medical robot is not one of autonomous agents, but of deeply integrated, cooperative systems that elevate the entire practice of medicine.
