In recent years, the increasing prevalence of peripheral nerve disorders, such as facial paralysis, sciatica, neuritis, and various nerve injuries, has posed significant challenges to global healthcare systems. These conditions often arise from poor lifestyle habits and suboptimal physical health, leading to severe impacts on patients’ quality of life. For cases where pharmacological treatments fail or when nerve damage is accompanied by bone or soft tissue injuries, minimally invasive surgery emerges as a critical therapeutic pathway. However, the complexity of peripheral nerve systems—characterized by diverse anatomical locations, intricate structures, and dynamic physiological changes—makes minimally invasive procedures exceptionally difficult. Key issues include real-time accurate positioning amid human body movements, the lack of flexible minimally invasive medical robot technologies for human-machine collaboration, and the inability to perform real-time interactive surgeries with precision. Additionally, existing minimally invasive instruments, typically with diameters around 5 mm, are often too large for micro-scale nerve lesions, necessitating the development of finer, more precise surgical devices. The absence of adaptive, self-adjusting minimally invasive techniques further complicates procedures, as minor tissue movements can disrupt planned trajectories, causing repeated surgeries and additional patient trauma. In this review, I will explore the advancements in medical robot technologies aimed at addressing these challenges, focusing on neural applications. The integration of medical robots into clinical practice has revolutionized minimally invasive surgery, enabling enhanced precision, dexterity, and safety. Throughout this discussion, I will emphasize the role of medical robots in transforming neural interventions, and I will incorporate tables and formulas to summarize key concepts and models.
The evolution of medical robots for neural minimally invasive surgery has been driven by the need to overcome limitations in traditional techniques. In many clinical settings, rigid handheld devices are still predominant, as shown in early implementations where fixed-angle instruments or CT-guided straight needles were used for procedures like sciatic nerve surgery. These approaches suffer from low real-time accuracy, high radiation exposure from CT, inability to treat complex lesion sites, and inefficiencies leading to repeated insertions and secondary damage. This highlights the urgent demand for adaptive medical robot systems that can navigate dynamic anatomical environments. The development of such medical robots involves interdisciplinary efforts, merging medicine, biomechanics, mechanical engineering, materials science, computer science, and robotics. These systems augment surgeons’ capabilities by providing visual, tactile, and auditory feedback, thereby improving diagnostic and therapeutic outcomes. In this review, I will analyze the progression from rigid to flexible medical robots, the integration of navigation technologies, and the future directions for this field. The term “medical robot” will be frequently referenced to underscore its centrality in advancing minimally invasive neural surgery.
In China, the adoption of medical robots for neural applications began in the mid-1990s, with early systems focusing on neurosurgery, orthopedics, and urology. For instance, initial research involved using industrial robot platforms for brain surgery positioning, demonstrating the potential of robotic assistance in enhancing surgical accuracy. Over time, collaborations between academic institutions and hospitals led to the creation of optical navigation-integrated medical robot systems for craniofacial surgery, aiming at precise treatment through controlled robotic movements. These early medical robots laid the groundwork for more sophisticated systems, but they often relied on rigid structures that limited adaptability in variable tissue environments. Further advancements included the development of puncture force modeling and soft tissue simulation for minimally invasive procedures, where experimental setups incorporated multi-sensor needle holders to study safety and positioning. However, these systems lacked real-time CT integration and adaptive path-changing capabilities, indicating gaps in flexible navigation for medical robots. In general surgery, innovations like the “Miaoshou” robot system enabled complex procedures such as gallbladder removal and colon tumor resection, while orthopedic medical robots allowed for precise implant placement in spinal surgeries. Despite these successes, the challenge of achieving millimeter-scale, flexible, and real-time navigated minimally invasive surgery with medical robots remained unaddressed, particularly for peripheral nerve interventions where tissue mobility and fine-scale lesions are critical.
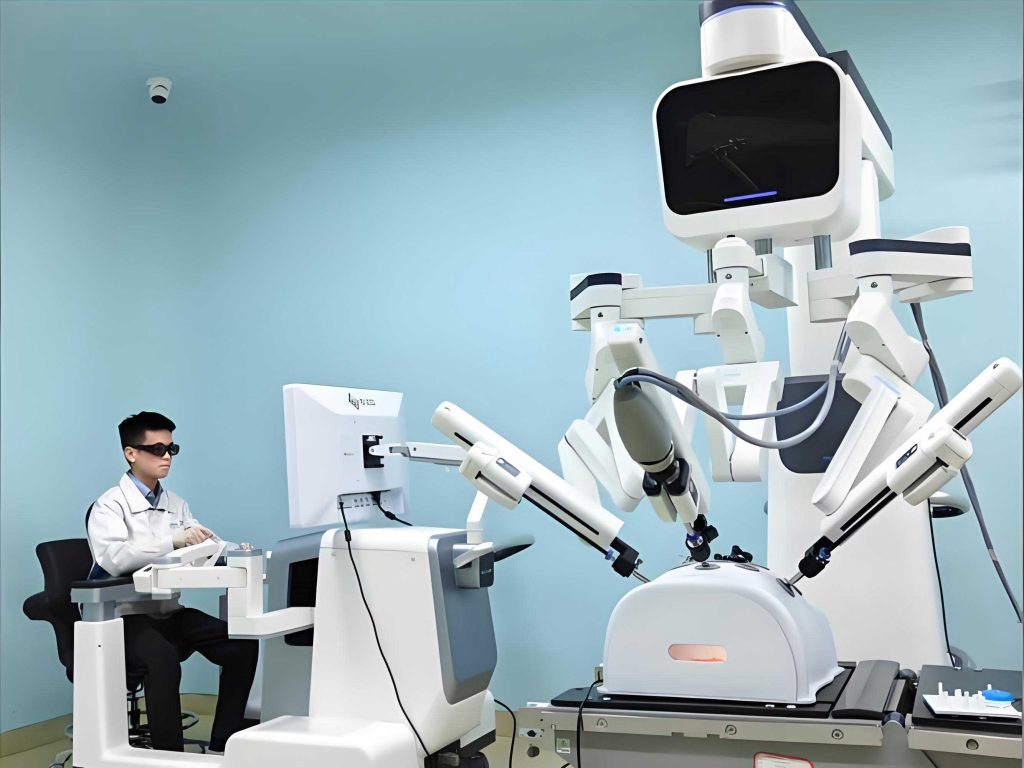
The transition to flexible minimally invasive medical robots represents a significant leap forward. Unlike collaborative medical robots that assist externally, flexible medical robots enter the body directly, requiring high dexterity, controllability, and miniaturization. International research, notably from teams in the Netherlands, has pioneered wire-driven flexible structures with diameters as small as 0.4-0.5 mm. These medical robots feature front-end flexible spring mechanisms, curved path feed systems, and hybrid rigid-flexible designs that enable navigation through complex anatomical pathways. They have been applied in diverse clinical scenarios, including mastitis, cholecystitis, and cyst removal, showcasing their versatility. However, these early flexible medical robots were primarily manually operated without intelligent navigation, limiting their autonomy. In contrast, systems like the da Vinci surgical medical robot have set benchmarks for human-machine collaboration in minimally invasive surgery. With components such as a surgeon console, bedside robotic arms, and 3D imaging, the da Vinci medical robot provides enhanced precision and magnification for procedures in gynecology and other fields. Yet, its reliance on rigid instruments with diameters around 5 mm restricts access to micro-scale lesions and requires larger operational spaces, underscoring the need for finer, more adaptable medical robots. Recent developments in China have introduced externally navigated millimeter-scale medical robots for neurosurgery, offering improved accuracy but still using rigid needles that lack flexibility for variable trajectories. Meanwhile, emerging autonomous flexible medical robots from the U.S. incorporate AI and micro-navigation for cardiac surgery, though clinical adoption and regulatory compliance are ongoing concerns. A collaborative effort in 2020 proposed a smart flexible wire-driven medical robot system with multi-dimensional visual navigation, aiming for sub-3 mm diameter applications in neural surgery, which highlights the trend toward integrating external and internal sensing for real-time control.
To better understand the technological landscape, I will summarize key aspects of medical robot systems in Table 1, comparing their characteristics, advantages, and limitations. This table illustrates the evolution from rigid to flexible designs and the integration of navigation features in medical robots.
| Medical Robot Type | Key Features | Diameter/Scale | Navigation Method | Applications | Limitations |
|---|---|---|---|---|---|
| Rigid Handheld Devices | Fixed-angle or straight needles, CT-guided | ~5 mm | CT imaging, manual control | Sciatic nerve surgery, basic biopsies | Low accuracy, high radiation, tissue damage |
| Early Collaborative Medical Robots | Industrial robot bases, optical navigation | Several cm | External sensors, pre-planning | Neurosurgery, orthopedics | Limited flexibility, no real-time adaptation |
| Da Vinci Medical Robot | Surgeon console, 3D vision, robotic arms | 5 mm instruments | 3D endoscopic imaging | Gynecology, general surgery | Large footprint, rigid tools, costly |
| Flexible Wire-Driven Medical Robots | Spring structures, wire control, hybrid design | 0.4-0.5 mm | Manual operation, basic visualization | Micro-scale lesions, complex pathways | Lack of autonomous navigation |
| Smart Flexible Medical Robots | AI integration, multi-sensor feedback, adaptive control | <3 mm | External/internal visual fusion | Peripheral nerve surgery, variable paths | Under development, regulatory hurdles |
The effectiveness of medical robots in neural minimally invasive surgery heavily depends on understanding the biomechanical interactions between the robot and tissue. When a medical robot needle or instrument penetrates muscle or nerve tissue, it encounters forces influenced by tissue viscosity, layered contractions, and transient local stimuli. These factors can deflect the needle from its intended path, reducing accuracy. To model this, I consider the puncture force dynamics, which can be expressed using a simplified equation that accounts for elastic, viscous, and frictional components. The total force \( F_{\text{total}} \) during insertion can be described as:
$$ F_{\text{total}} = F_{\text{elastic}} + F_{\text{viscous}} + F_{\text{friction}} $$
where \( F_{\text{elastic}} \) represents the tissue’s elastic resistance, often modeled as \( F_{\text{elastic}} = k \cdot x \), with \( k \) being the tissue stiffness and \( x \) the displacement. The viscous force \( F_{\text{viscous}} \) relates to the velocity of insertion, given by \( F_{\text{viscous}} = c \cdot v \), where \( c \) is the damping coefficient and \( v \) is the velocity. Frictional force \( F_{\text{friction}} \) depends on the contact interface, expressed as \( F_{\text{friction}} = \mu \cdot N \), with \( \mu \) as the friction coefficient and \( N \) as the normal force. In practice, these parameters vary across tissue types and physiological states, necessitating adaptive control in medical robots. For instance, a medical robot might use real-time force feedback to adjust its trajectory, minimizing deviation. This can be formulated as a control law:
$$ u(t) = K_p \cdot e(t) + K_i \cdot \int e(t) dt + K_d \cdot \frac{de(t)}{dt} $$
where \( u(t) \) is the control input to the medical robot actuators, \( e(t) \) is the error between desired and actual position, and \( K_p \), \( K_i \), \( K_d \) are proportional, integral, and derivative gains, respectively. Such models underscore the complexity of integrating mechanical design with intelligent algorithms in medical robots.
Visual navigation is another cornerstone of advanced medical robots. For neural applications, combining external imaging (e.g., CT, MRI) with internal micro-visual feedback enables precise localization of millimeter-scale lesions. The fusion of these data streams can be represented as an optimization problem. Let \( P_{\text{ext}} \) denote the position from external navigation (e.g., based on preoperative scans), and \( P_{\text{int}} \) denote the position from internal sensors (e.g., micro-cameras or fiber optics on the medical robot). The estimated true position \( P_{\text{true}} \) can be obtained by minimizing a cost function:
$$ P_{\text{true}} = \arg \min_{P} \left( \alpha \| P – P_{\text{ext}} \|^2 + \beta \| P – P_{\text{int}} \|^2 \right) $$
where \( \alpha \) and \( \beta \) are weighting factors reflecting the reliability of each navigation source. In smart medical robots, machine learning techniques may further refine this estimation by learning from historical data. For example, a neural network could predict tissue deformation during insertion, enhancing the medical robot’s path-planning capability. The integration of such multi-scale visual systems allows medical robots to adapt to dynamic anatomical changes, crucial for procedures like nerve decompression or biopsy in mobile tissues.
Flexible medical robot design poses unique challenges in materials and actuation. To achieve multi-degree-of-freedom (DOF) steering in confined spaces, many medical robots employ tendon-driven mechanisms or shape-memory alloys. The kinematics of a flexible medical robot segment can be modeled using continuum mechanics approaches. Consider a flexible rod with length \( L \) and curvature \( \kappa(s) \) along its arc length \( s \). The tip position \( \mathbf{r}(L) \) can be derived from:
$$ \mathbf{r}(L) = \int_0^L \mathbf{R}(s) \cdot \mathbf{e}_z \, ds $$
where \( \mathbf{R}(s) \) is the rotation matrix describing the orientation along the rod, and \( \mathbf{e}_z \) is the local tangent vector. For a tendon-driven medical robot with \( n \) tendons, the curvature is related to tendon displacements \( \Delta l_i \) by:
$$ \kappa = \frac{\sum_{i=1}^n w_i \Delta l_i}{L \cdot r} $$
where \( w_i \) are weighting factors and \( r \) is the distance from the tendon to the central axis. This enables precise control of bending for navigation around nerves. Material selection is critical; polymers like silicone or polyurethane offer flexibility, while embedded sensors (e.g., fiber Bragg gratings) provide strain feedback. The mechanical properties can be summarized in Table 2, which compares materials used in flexible medical robots.
| Material | Young’s Modulus (MPa) | Flexibility | Biocompatibility | Typical Use in Medical Robots |
|---|---|---|---|---|
| Silicone Rubber | 0.5-5 | High | Excellent | Outer sheath, sealing |
| Polyurethane | 10-100 | Moderate | Good | Structural layers |
| Nitinol (Shape Memory Alloy) | 30-80 | Variable | Good | Actuators, steerable tips |
| Polyimide | 2000-3000 | Low | Good | Printed circuit boards |
| Hydrogels | 0.01-1 | Very High | Excellent | Emerging for soft robotics |
The control architecture of a medical robot system often involves hierarchical layers: high-level planning based on medical images, mid-level trajectory generation, and low-level actuator control. For neural applications, safety is paramount, so redundancy and fail-safe mechanisms are incorporated. A typical control loop for a medical robot might include impedance control to manage tissue interaction forces, described by:
$$ \mathbf{F} = \mathbf{M} \ddot{\mathbf{x}} + \mathbf{B} \dot{\mathbf{x}} + \mathbf{K} \mathbf{x} $$
where \( \mathbf{F} \) is the force vector, \( \mathbf{x} \) is the position vector, and \( \mathbf{M} \), \( \mathbf{B} \), \( \mathbf{K} \) are inertia, damping, and stiffness matrices, respectively. By adjusting these matrices, the medical robot can exhibit compliant behavior, reducing the risk of tissue damage. Moreover, haptic feedback systems can transmit forces sensed by the medical robot to the surgeon, enhancing teleoperation. This is especially useful in delicate nerve surgeries where tactile cues are vital.
Looking forward, several key technologies must be advanced to realize the full potential of medical robots in neural minimally invasive surgery. First, deeper insights into biomechanical and dynamical tissue properties are needed. The influence of muscle viscoelasticity, multi-layer contractions, and transient stimuli on medical robot needle insertion must be quantified through combined computational and experimental studies. This will inform the design of adaptive medical robot structures that compensate for tissue motion. Second, flexible medical robot architectures must be optimized for millimeter-scale operations. This involves developing series of miniaturized, multi-DOF instruments with integrated sensing, enabling variable-path precision in diverse anatomical regions. Third, multi-scale visual navigation systems require enhancement. Fusion of real-time internal micro-visual data with external imaging (e.g., ultrasound or low-dose CT) can reduce radiation exposure and improve targeting accuracy for sub-millimeter lesions. Finally, smart control algorithms leveraging AI and machine learning can enable autonomous or semi-autonomous functions in medical robots, such as real-time path replanning and anomaly detection. However, ethical and regulatory frameworks must evolve alongside these technological advancements to ensure safety and efficacy.
In conclusion, the field of medical robots for neural minimally invasive surgery is rapidly evolving, driven by the need for precision, flexibility, and adaptability. From early rigid systems to contemporary flexible designs, medical robots have demonstrated significant improvements in surgical outcomes. The integration of advanced navigation, smart materials, and intelligent control holds promise for addressing the complex challenges of peripheral nerve interventions. As research progresses, I anticipate that next-generation medical robots will become more pervasive in clinical settings, offering tailored solutions for micro-scale pathologies and enhancing patient care. Continued interdisciplinary collaboration will be essential to overcome technical hurdles and translate innovations into practical medical robot applications, ultimately benefiting society through reduced trauma and faster recovery times.
To encapsulate the discussed technologies, I present a summary of key equations and models relevant to medical robots in Table 3. This table serves as a quick reference for the mathematical foundations underlying medical robot design and control.
| Concept | Equation | Description | Application in Medical Robots |
|---|---|---|---|
| Puncture Force Dynamics | \( F_{\text{total}} = kx + cv + \mu N \) | Models tissue resistance during insertion | Force feedback control for accurate needle steering |
| PID Control Law | \( u(t) = K_p e(t) + K_i \int e(t) dt + K_d \frac{de(t)}{dt} \) | Standard control for error correction | Actuator control in medical robot systems |
| Navigation Fusion | \( P_{\text{true}} = \arg \min_{P} \left( \alpha \| P – P_{\text{ext}} \|^2 + \beta \| P – P_{\text{int}} \|^2 \right) \) | Optimizes position estimation from multiple sources | Real-time localization in medical robots |
| Flexible Rod Kinematics | \( \mathbf{r}(L) = \int_0^L \mathbf{R}(s) \cdot \mathbf{e}_z \, ds \) | Describes tip position of continuum robot | Path planning for flexible medical robots |
| Impedance Control | \( \mathbf{F} = \mathbf{M} \ddot{\mathbf{x}} + \mathbf{B} \dot{\mathbf{x}} + \mathbf{K} \mathbf{x} \) | Governs interaction forces with environment | Safe tissue manipulation by medical robots |
The journey toward ideal medical robots for neural surgery is ongoing, but with each technological breakthrough, we move closer to realizing minimally invasive procedures that are both precise and gentle. As I reflect on the advancements, it is clear that medical robots will continue to play a pivotal role in shaping the future of healthcare, offering new hope for patients with challenging nerve disorders.
