The surgical management of hypertensive intracerebral hemorrhage (HICH) has long presented a formidable challenge in neurosurgery. As a neurosurgeon, my primary goals are to evacuate the hematoma to relieve mass effect, minimize secondary neuronal injury, and preserve neurological function—all while inflicting the least possible iatrogenic trauma. Traditional approaches, from large craniotomies to frame-based stereotaxy, have inherent limitations that directly impact patient outcomes. The former, while offering direct visualization, is inherently invasive. The latter, though more precise, involves the application of a rigid head frame, a process that can be painful and provoke hypertensive surges, ironically increasing the risk of the very complication we seek to treat: re-bleeding.
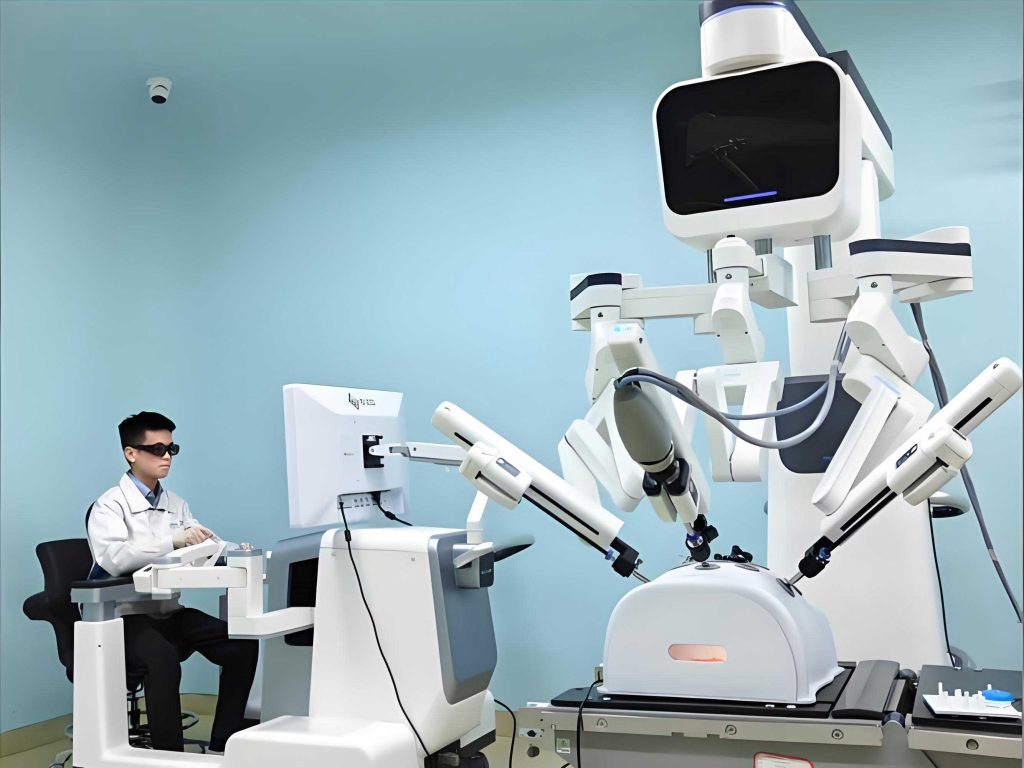
This narrative chronicles a significant evolution in my practice: the integration of a domestic, frameless stereotactic medical robot into the treatment paradigm for HICH. This shift is not merely a change of tools; it represents a fundamental advancement towards minimally invasive, maximally precise surgery. The core of this approach is the synergy between advanced imaging, computational planning, and the unwavering mechanical accuracy of a robotic arm. The medical robot transforms pre-operative CT data into a dynamic, three-dimensional roadmap, allowing for surgical planning with an accuracy measured in sub-millimeters. It enables trajectories that are optimized for the hematoma’s unique morphology while meticulously avoiding eloquent brain structures and vascular territories, a level of planning fidelity difficult to achieve consistently with manual techniques.
The evolution of the medical robot in this context is a response to the critical need for reducing procedural morbidity. The transition from framed to frameless systems eliminated a significant source of patient discomfort and physiological stress. Now, with robotic integration, we have moved beyond simple navigation to active, steady instrument guidance. The robot’s arm acts as an ultra-stable, “third hand” that precisely aligns to the pre-planned trajectory and holds that position indefinitely without tremor. This stability is crucial when navigating through narrow corridors of brain tissue to reach a deep-seated hematoma. The role of the medical robot is thus multifaceted: it is a planner, a navigator, and a precise mechanical guide, all controlled from a single workstation.
Pathophysiology and Clinical Imperatives in HICH
Understanding HICH is key to appreciating the value of robotic intervention. It typically originates from the rupture of small, perforating arteries in deep brain structures—the basal ganglia, thalamus, pons, and cerebellum—that have been weakened by chronic hypertension and lipohyalinosis. The initial hemorrhage creates a mass lesion, but the injury cascade extends far beyond this primary event.
- Primary Injury: The physical disruption of tissue and immediate mass effect.
- Secondary Injury: A complex, hours-to-days long process involving peri-hematomal edema, inflammation, cytotoxicity from blood breakdown products (like thrombin and hemoglobin), and local ischemia.
The volume and location of the hematoma are the most critical prognostic factors. The widely referenced surgical thresholds—approximately 30 mL for supratentorial and 10 mL for infratentorial hemorrhages—serve as general guides. However, the decision to operate is nuanced. A smaller hematoma (e.g., 20-25 mL) situated in the posterior limb of the internal capsule can cause devastating hemiparesis, while a larger one in a relatively silent area may be better tolerated. The equation for surgical decision-making incorporates not just volume, but also neurological deficit, signs of herniation, and the patient’s overall condition. The fundamental surgical principle is to evacuate the clot efficiently to halt the secondary injury cascade and decompress neural pathways, creating the optimal biological environment for recovery.
The traditional surgical dilemma has been balancing the aggressiveness of evacuation with the trauma of access. Large craniotomies achieve good evacuation but with significant collateral damage. Simple bedside catheter placements are minimally invasive but often blind and imprecise, risking inadequate drainage or new injury. This is precisely the gap that robot-assisted surgery aims to bridge.
The Robotic Surgical System: Architecture and Workflow
The medical robot system used in this evolution is a sophisticated integration of hardware and software. Its core components and their functions can be summarized as follows:
| System Component | Description | Function in HICH Surgery |
|---|---|---|
| Robotic Manipulator Arm | A multi-jointed, 6-degree-of-freedom mechanical arm. | Provides stable, precise positioning and guidance for surgical instruments along the pre-planned trajectory. Its flexibility allows access from various angles. |
| Optical Localization System | Stereo cameras or infrared sensors. | Tracks fiducial markers on the patient’s head, registering the patient’s physical space to the pre-operative image space with high accuracy. |
| Planning & Control Station | Computer workstation with dedicated software. | Used for image import, 3D reconstruction, hematoma segmentation, trajectory planning, and controlling the robotic arm. |
| Patient Interface & Fixation | Molded headrest or similar non-invasive clamp. | Secures the patient’s head during surgery without the need for a painful invasive frame, enhancing comfort and safety. |
The surgical workflow is a structured, stepwise process that maximizes safety and precision:
- Pre-operative Imaging and Planning: A high-resolution CT scan is performed with 4-5 fiducial markers affixed to the patient’s scalp. The DICOM data is loaded into the medical robot‘s software. The hematoma is segmented, and its volume can be calculated using the ellipsoid formula approximation:
$$ V = \frac{ABC}{2} $$
where \(A\), \(B\), and \(C\) are the orthogonal diameters of the hematoma. The surgeon then plans the optimal trajectory. Key planning principles include:- Targeting the geometric center or the largest cross-sectional area of the hematoma.
- Designing the entry point and path to avoid sulci, major vessels (like the Sylvian fissure), and eloquent cortex.
- Planning for single or multiple trajectories based on hematoma shape (e.g., a bilobed clot may benefit from two catheters).
- Registration and Draping: In the OR, the patient’s head is secured with a non-invasive head holder. The medical robot is positioned and its arm is “registered” by having its cameras detect the fiducial markers. This creates a one-to-one mapping between the virtual plan and the real-world patient. The system’s accuracy, often reported as Target Registration Error (TRE), is a critical metric:
$$ TRE = \sqrt{(x_{actual} – x_{planned})^2 + (y_{actual} – y_{planned})^2 + (z_{actual} – z_{planned})^2} $$
For a clinical-grade medical robot, this error is typically less than 1.5 mm. - Robotic Guidance and Procedure: Under local anesthesia, the robotic arm autonomously moves to the planned entry point. A small stab incision and a mini-burr hole (often just 2-3 mm) are made. The arm then guides a cannula or aspiration needle along the exact pre-planned path to the target. The surgeon performs aspiration, often removing 50-70% of the liquid component of the clot. A soft drainage catheter is then placed down the same trajectory.
- Post-operative Management: CT confirms catheter position and initial evacuation. The catheter is then used for closed-system drainage, often with instillation of a thrombolytic agent (e.g., 20,000-50,000 IU of urokinase or 1 mg of rt-PA) every 8-12 hours to liquefy and drain the remaining clot over 2-3 days.
Comparative Analysis: Robotic vs. Conventional Approaches
The advantages of the medical robot-assisted approach become stark when compared to traditional methods. The following table synthesizes key comparative metrics:
| Parameter | Conventional Craniotomy | Frame-Based Stereotaxy | Freehand/Manual CT-Guided Drainage | Robotic-Assisted Stereotaxy |
|---|---|---|---|---|
| Invasiveness | High (large bone flap, brain retraction) | Moderate (frame pins, single burr hole) | Low (single burr hole) | Very Low (mini-burr hole, no frame) |
| Precision & Planning | Direct vision, limited pre-planning | High, but trajectory options limited by frame arc | Low, dependent on surgeon’s mental calculation | Very High, with unlimited 3D trajectory planning |
| Procedure Time (Setup + Surgery) | Long | Moderate to Long (frame application) | Short | Moderate (planning takes time, execution is fast) |
| Patient Comfort | Low (post-operative pain) | Low (frame application pain) | High | High |
| Adaptability to Clot Shape | Good under direct vision | Fair (single linear trajectory) | Poor | Excellent (multi-trajectory planning possible) |
| Learning Curve | Steep | Moderate | Shallow but outcome-variable | Moderate (requires comfort with software) |
From a clinical outcomes perspective, the precision of the medical robot translates into tangible benefits. The ability to place the catheter optimally within the hematoma leads to more efficient evacuation. A key metric is the rate of hematoma resolution over time. Let \( V_0 \) be the initial hematoma volume, \( V_s \) be the volume immediately after surgery, and \( V_t \) be the volume at time \( t \) post-thrombolysis. The evacuation efficiency \( \eta \) of the initial procedure can be defined as:
$$ \eta = \frac{V_0 – V_s}{V_0} \times 100\% $$
With robotic guidance, \( \eta \) is consistently higher and more predictable than with manual methods. Furthermore, the reduction in operative trauma and the precision in avoiding critical structures theoretically lowers the rate of complications such as new neurological deficits, infections, and symptomatic re-bleeding. While large randomized trials are still needed, cohort studies suggest a trend towards improved functional outcomes (e.g., higher rates of achieving a modified Rankin Scale score of 0-3 at 6 months) with minimally invasive approaches, with robotics offering the most refined version of this technique.
Expanding the Indications: The Case for Smaller, Strategic Hematomas
One of the most profound implications of robotic precision is the re-evaluation of surgical indications. Classically, smaller hematomas (<30 mL) were often managed conservatively. However, a 25 mL clot in the internal capsule can destroy the corticospinal tract, leading to permanent, severe hemiplegia. The low morbidity of robotic access changes the risk-benefit calculus dramatically.
The argument for early intervention in these cases is based on the pathophysiology of pressure and ischemia. The clot exerts a localized pressure \( P_{clot} \) on adjacent white matter tracts. This pressure, combined with toxic metabolites, creates a zone of impaired function and threatened viability. The time \( T \) for which this pressure is applied is directly related to the probability of irreversible axonal injury \( P_{irr} \), a relationship that can be conceptually modeled as:
$$ P_{irr} \propto \int_{0}^{T} (P_{clot}(t) + C_{tox}(t)) \, dt $$
where \( C_{tox}(t) \) represents the concentration of cytotoxic agents. Early robotic evacuation rapidly reduces \( P_{clot} \) and facilitates the clearance of \( C_{tox} \), thereby minimizing the integral and preserving neural function. Therefore, for strategically located hematomas causing significant deficit, the medical robot enables a proactive, preventative surgical strategy rather than a salvage procedure. The goal shifts from merely saving life to actively preserving and restoring neurological quality of life.
Technical Nuances, Complications, and Managing Expectations
While powerful, the medical robot is not a magical solution. Its success hinges on meticulous technique and awareness of pitfalls. A key intra-operative decision is planning single versus multiple trajectories. A single catheter along the long axis of a sausage-shaped hematoma is often sufficient. For irregular, bilobed, or very large clots, two catheters can be planned to create a more efficient drainage “network.” The software allows for simulating the effective drainage radius of each catheter.
| Complication | Potential Cause | Robotic & Technical Mitigation |
|---|---|---|
| Re-bleeding | Injury to a vessel during trajectory; underlying coagulopathy; postoperative hypertension. | Careful trajectory planning to avoid vascular sulci and known vessels. Strict peri-operative BP control. Post-op CT protocol. The robot’s precision itself reduces the risk of stray vessel injury. |
| Inadequate Drainage | Malpositioned catheter; catheter clogging; insufficient thrombolysis. | Precise robotic placement ensures optimal initial position. Use of larger-bore soft catheters. Protocolized thrombolytic regimen with regular catheter flushing. |
| Infection | Break in sterile technique; prolonged catheter indwelling time. | Meticulous draping of the robotic arm. Limiting drainage to 3-5 days. Antibiotic protocol. |
| Registration Failure/Error | Movement of fiducial markers; software error. | Secure fiducial placement. System redundancy checks. Always verifying registration accuracy on a known landmark before proceeding. |
A critical lesson is that the medical robot is a tool for precise access, not a treatment for underlying disease. It cannot correct a severe coagulopathy. A patient with an undiagnosed platelet function disorder or refractory hypertension may re-bleed despite perfect catheter placement. Therefore, the robotic procedure must be embedded within comprehensive neurological and medical intensive care.
Economic and Broader Clinical Implications
The adoption of a medical robot system represents a significant capital investment. A full cost-effectiveness analysis must consider direct costs (device, disposables), indirect costs (OR time, maintenance), and set them against outcomes. The formula for a simplified cost-benefit ratio \( R \) over a patient cohort could be considered as:
$$ R = \frac{C_{robot} + C_{complications, robot}}{C_{standard} + C_{complications, standard}} $$
where \( C \) represents costs and the denominator includes the often-high costs of managing complications (re-operation, extended ICU stay, long-term rehabilitation). While \( C_{robot} \) is high, the evidence suggests that \( C_{complications, robot} \) and long-term rehabilitation costs may be lower due to better outcomes. Reduced length of stay in the ICU and acute ward is a major potential driver of cost savings. Furthermore, the versatility of the medical robot—its use in deep brain stimulation (DBS) lead placement, stereo-EEG implantation, and brain biopsy—allows the fixed cost to be amortized across multiple neurosurgical services, improving its overall economic viability for a department.
Beyond economics, the proliferation of domestic robotic systems has important implications for global healthcare access. Internationally sourced systems can be prohibitively expensive for many hospitals. The development and validation of effective domestic medical robot platforms can lower the barrier to entry, making this high-precision care available to a much larger patient population, potentially standardizing and elevating the quality of minimally invasive neurosurgery on a wide scale.
Future Directions and Conclusion
The journey with the medical robot in HICH surgery is just beginning. Future developments point towards even greater integration and intelligence:
- Real-Time Imaging Integration: Combining the robotic system with intra-operative CT or ultrasound would allow for dynamic updating of the plan, accounting for brain shift during drainage and confirming evacuation in real time.
- Enhanced Haptic and Force Feedback: Future robotic systems may provide the surgeon with sensitive feedback during aspiration, helping to distinguish between clot and brain tissue.
- AI-Powered Planning: Machine learning algorithms could analyze thousands of prior cases to suggest the optimal number of trajectories, target points, and even predict the risk of complications for a specific hematoma morphology.
- Tele-Robotic Capabilities: In principle, a medical robot could be guided remotely by a specialist, bringing expert-level minimally invasive care to underserved or remote locations.
In conclusion, the integration of robotic technology into the surgical management of hypertensive intracerebral hemorrhage marks a paradigm shift. It successfully addresses the historical trade-off between invasiveness and precision. The medical robot provides a stable, accurate, and minimally invasive conduit to deep-seated brain pathology. It empowers surgeons to execute complex pre-operative plans with sub-millimeter fidelity, improving the efficiency of hematoma evacuation while minimizing collateral damage. Perhaps most importantly, its low morbidity profile encourages earlier intervention for strategically damaging smaller hematomas, shifting the therapeutic goal from salvage to preservation of neurological function. As the technology evolves and becomes more accessible, the medical robot is poised to become a cornerstone in the modern neurosurgeon’s arsenal for treating one of neurology’s most devastating conditions, offering patients a path to recovery that is less burdensome and more hopeful than ever before.
