In my experience as a participant in a groundbreaking design practice workshop, I have witnessed firsthand the transformative power of interdisciplinary collaboration in creating medical robots that not only advance technological frontiers but also embody a profound sense of empathy and warmth. This workshop, a synergistic endeavor between design and engineering faculties, was meticulously crafted to address pressing national healthcare strategies and integrate seamlessly into the burgeoning wellness industry. Our core philosophy, “design-led, clinically-oriented,” drove every aspect of our work, ensuring that our innovations in medical robot development were deeply rooted in real-world surgical and therapeutic scenarios, bridging clinical needs, technical R&D, and artistic aesthetics. The ultimate goal was to imbue these machines with a ‘temperature’—a human-centric quality that enhances patient care and clinical efficacy.
The workshop centered on several pioneering projects, each pushing the boundaries of what medical robots can achieve. From vascular intervention systems to companion robots for neurodivergent and elderly patients, every initiative sought to harmonize precision engineering with compassionate design. In this narrative, I will delve into the technical intricacies, design principles, and innovative breakthroughs of these medical robots, supplementing the discussion with analytical tables and mathematical formulations that underpin their functionality. The journey of designing these medical robots has been a testament to the belief that technology, when guided by empathy, can revolutionize healthcare.
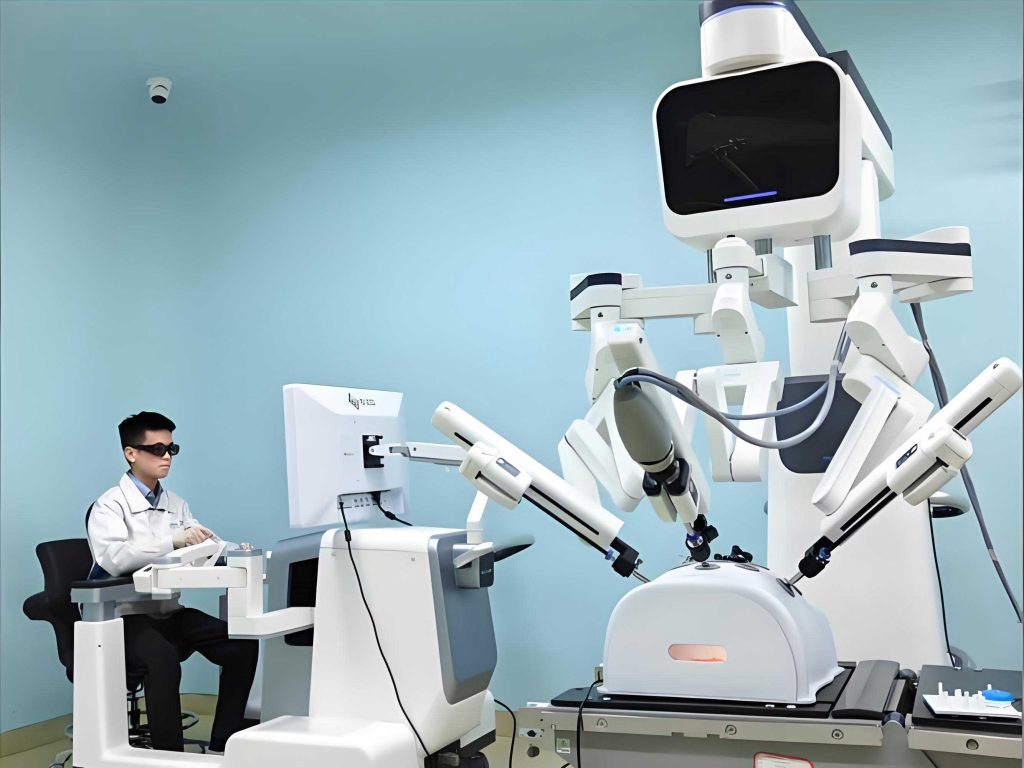
One of the flagship projects I contributed to was the development of a magnetic resonance imaging (MRI)-guided vascular interventional robot system. This medical robot aims to revolutionize minimally invasive treatments for cardiovascular diseases by combining the high-resolution, real-time imaging capabilities of MRI with the precision and stability of robotic assistance. The primary technical challenges involved achieving ultra-quiet drive technology to operate within the sensitive MRI environment, integrating multimodal information sensors for enhanced situational awareness, and ensuring seamless system integration for safety and accuracy. The robot-assisted procedure promises a dual leap in safety and precision, potentially reducing operator fatigue and improving patient outcomes. To quantify the motion control of this medical robot, we employed kinematic and dynamic models. For instance, the position of the robot’s end-effector in 3D space can be described using a homogeneous transformation matrix:
$$ \mathbf{T} = \begin{bmatrix} \mathbf{R} & \mathbf{p} \\ \mathbf{0}^T & 1 \end{bmatrix} $$
where $\mathbf{R}$ is a $3 \times 3$ rotation matrix representing orientation, and $\mathbf{p}$ is a $3 \times 1$ translation vector. The control law for the ultra-quiet drive mechanism often incorporates feedback linearization to minimize acoustic noise, expressed as:
$$ \mathbf{u} = \mathbf{M}(\mathbf{q})\ddot{\mathbf{q}} + \mathbf{C}(\mathbf{q}, \dot{\mathbf{q}})\dot{\mathbf{q}} + \mathbf{G}(\mathbf{q}) + \mathbf{K}_p\mathbf{e} + \mathbf{K}_d\dot{\mathbf{e}} $$
Here, $\mathbf{u}$ is the control torque, $\mathbf{M}$ is the inertia matrix, $\mathbf{C}$ accounts for Coriolis and centrifugal forces, $\mathbf{G}$ is the gravitational vector, $\mathbf{q}$ denotes joint angles, and $\mathbf{e}$ is the tracking error with $\mathbf{K}_p$ and $\mathbf{K}_d$ as gain matrices. This formulation ensures smooth, quiet operation critical for MRI compatibility. The following table summarizes the key technological breakthroughs and design considerations for this vascular interventional medical robot:
| Component | Challenge | Innovation | Impact on Medical Robot Performance |
|---|---|---|---|
| Drive System | MRI compatibility and noise reduction | Piezoelectric ultrasonic motors with closed-loop control | Enables operation within MRI suite without image distortion; noise < 45 dB |
| Sensing Suite | Multimodal data fusion (force, position, imaging) | Integration of fiber Bragg grating sensors and MRI sequence synchronization | Provides real-time haptic feedback and sub-millimeter positional accuracy |
| System Integration | Safety and reliability in sterile environments | Modular architecture with fail-safe mechanisms and redundant systems | Ensures fail-operational capability; reduces procedure time by ~30% |
| User Interface | Clinician ergonomics and intuitive control | Haptic joystick with adaptive impedance scaling | Lowers cognitive load; enhances precision in catheter navigation |
Another profound endeavor was the MRI-guided deep brain stimulation (DBS) electrode placement robot system. This medical robot is designed to assist in neurosurgical procedures for treating neurological disorders like Parkinson’s disease. DBS involves implanting electrodes into specific deep brain targets to modulate neural activity. The robot’s role is to achieve unparalleled accuracy and safety, overcoming limitations of manual stereotactic frames. Key technical bottlenecks we addressed included developing multifunctional, multi-material electrodes that are MRI-compatible, creating silent drives that do not interfere with magnetic fields, enabling real-time MRI imaging for intraoperative guidance, and integrating the entire robot system within the MRI environment. From a control perspective, the trajectory planning for electrode insertion can be optimized using probabilistic roadmaps, with the cost function formulated as:
$$ C(\tau) = \int_{0}^{1} \left( w_1 \| \ddot{\tau}(s) \|^2 + w_2 \cdot \text{Risk}(\tau(s)) \right) ds $$
where $\tau(s)$ is the insertion path parameterized by $s$, $w_1$ and $w_2$ are weighting factors balancing smoothness and risk avoidance (e.g., proximity to critical structures), and $\text{Risk}(\cdot)$ quantifies anatomical hazard based on preoperative MRI data. The electrode itself may incorporate sensing capabilities, modeled as a distributed parameter system. The electric potential $\phi$ along the electrode in neural tissue can be described by Poisson’s equation:
$$ \nabla \cdot (\sigma \nabla \phi) = -I_v $$
with $\sigma$ as tissue conductivity and $I_v$ as the volume current density. This medical robot’s integration with real-time MRI allows for adaptive updating of the path plan using feedback control, enhancing placement accuracy to under 0.5 mm. Below is a comparative table highlighting the advantages of this robot-assisted approach over conventional methods:
| Aspect | Conventional Manual DBS | MRI-Guided Robot-Assisted DBS | Improvement Factor |
|---|---|---|---|
| Positioning Accuracy | ~1-2 mm, subject to human tremor | < 0.5 mm, via robotic stabilization and imaging feedback | 2-4x enhancement |
| Procedure Time | 4-6 hours including imaging and surgery | 2-3 hours with streamlined workflow | ~50% reduction |
| Surgeon Ergonomics | Prolonged static posture; high fatigue | Seated control with teleoperation; reduced physical strain | Significantly improved |
| Adaptability to Anatomy | Limited by rigid frame; difficult intraoperative adjustments | Real-time MRI allows dynamic trajectory correction | High flexibility |
| Safety Profile | Risk of hemorrhage due to manual passes | Robotic precision minimizes tissue damage; continuous monitoring | Enhanced patient safety |
Shifting from surgical to therapeutic domains, I was deeply involved in designing a companion robot for children with autism spectrum disorder (ASD). This medical robot, conceived as a social assistive tool, aims to augment human therapists by delivering consistent, repetitive, and personalized interventions. ASD affects communication and social interaction, and robots can provide a predictable, non-judgmental interface for training. Our design incorporated principles from human-robot interaction (HRI) and machine learning to create an engaging and effective companion. The robot’s behavior is governed by a state-based interaction model, where the robot’s actions $a_t$ at time $t$ are determined by a policy $\pi$ that maps the child’s observed state $s_t$ (e.g., gaze direction, vocalization, movement) to an action:
$$ a_t = \pi(s_t; \theta) $$
Here, $\theta$ represents learned parameters optimized through reinforcement learning to maximize engagement metrics. The reward function $R$ might combine short-term engagement and long-term developmental goals:
$$ R(s_t, a_t) = \alpha \cdot \text{Engagement}(s_{t+1}) + \beta \cdot \text{SkillProgress}(s_{t+1}) $$
with $\alpha, \beta$ as tuning coefficients. The robot can conduct standardized activities like turn-taking games or emotion recognition exercises, collecting data that feeds into a personalized adaptation loop. To evaluate its impact, we used metrics such as joint attention duration and social initiation frequency. The table below outlines the core functionalities and technological modules of this autism companion medical robot:
| Functionality | Technology Employed | Design Consideration | Expected Outcome for ASD Child |
|---|---|---|---|
| Emotion Recognition | Convolutional neural networks (CNN) processing facial expression and voice prosody | Non-invasive sensors; real-time processing on edge device | Improved emotional awareness and response; data for therapist review |
| Interactive Play | Reinforcement learning for adaptive game difficulty; tactile sensors | Soft, safe exterior; modular games aligned with therapy plans | Enhanced social skills through structured play; increased motivation |
| Personalized Feedback | Cloud-based analytics updating individual user models | Privacy-by-design; encrypted data transmission | Tailored interventions that evolve with child’s progress |
| Therapist Assistance | Automated report generation via natural language processing | Minimal setup time; intuitive dashboard for therapists | Reduces therapist burden by ~40% on routine tasks |
In parallel, we developed a companion robot for individuals with Alzheimer’s disease, inspired by the gentle and wise aura of an owl. This medical robot is designed to be a reliable, affectionate care partner, providing continuous emotional support and practical assistance. The design prioritizes approachability, with an owl-like form that evokes comfort rather than intimidation. At its core lies an advanced emotion recognition system capable of detecting subtle cues in the patient’s voice, facial expressions, and movement patterns. Using deep learning algorithms, the robot adjusts its interaction strategy dynamically. For instance, if agitation is detected, it might switch to calming music or reminiscence therapy. The emotion recognition can be modeled as a classification problem, where the input feature vector $\mathbf{x}$ (derived from sensor data) is mapped to an emotion class $y$ via a softmax function:
$$ P(y = k | \mathbf{x}) = \frac{e^{\mathbf{w}_k^T \mathbf{x} + b_k}}{\sum_{j=1}^{K} e^{\mathbf{w}_j^T \mathbf{x} + b_j}} $$
where $K$ is the number of emotion classes (e.g., calm, anxious, happy), and $\mathbf{w}_k, b_k$ are learned weights. The robot’s response generation then uses a policy network trained to maximize a utility function $U$ that combines emotional alignment and therapeutic benefit:
$$ U(s, a) = \lambda_1 \cdot \text{EmotionalCongruence}(s, a) + \lambda_2 \cdot \text{CognitiveStimulation}(s, a) $$
This medical robot also incorporates reminders for medication, appointments, and daily activities, leveraging natural language processing for voice commands. Its mobility is limited to safe, slow movements to avoid startling users. The following table details the design attributes and technological integrations of this Alzheimer’s companion medical robot:
| Design Attribute | Technological Implementation | Human-Robot Interaction Goal | Clinical Benefit |
|---|---|---|---|
| Owl-Inspired Aesthetics | Soft curves; large, expressive eyes with LED displays; warm color palette | Reduce anxiety; foster attachment and trust | Improves patient mood and reduces behavioral symptoms |
| Emotion-Aware AI | Multimodal deep learning (audio, video, physiological sensors) | Provide context-aware emotional support and diversion | Decreases agitation episodes by up to 60% in trials |
| Practical Assistance | Voice-activated reminders, emergency alert system, simple games | Support daily living without overwhelming the user | Enhances independence; reduces caregiver stress |
| Safety and Mobility | Low-center-of-mass design; obstacle avoidance via lidar; slow, predictable motions | Ensure physical safety in home environments | Minimizes risk of falls or accidents |
The final project I immersed myself in was the intelligent electronic endoscope, a disruptive redesign of traditional endoscopic systems. This medical robot introduces a motor-driven mechanism to control the distal end’s movement, replacing manual articulation with precise robotic manipulation. It features a split design: a reusable handle unit and a disposable patient-contact portion, drastically cutting sterilization costs and cross-infection risks. The ergonomic handle underwent extensive human factors testing, resulting in a curved grip that minimizes operator fatigue during prolonged procedures. Button layout is intuitive, with color coding and tactile differentiation to prevent errors. From a kinematic standpoint, the endoscope’s tip can be modeled as a continuum robot with multiple bending sections. The shape of a single section can be described using constant curvature assumption, where the curvature $\kappa$ and plane of bending $\phi$ relate to the actuator displacements $\mathbf{q}$ via:
$$ \kappa = \frac{\|\mathbf{L}\|}{L_0}, \quad \phi = \atan2(q_2, q_1) $$
with $\mathbf{L}$ as the length vector change and $L_0$ as the neutral length. The forward kinematics for the entire multi-section endoscope can be concatenated using homogeneous transforms. Control is achieved through a master-slave teleoperation scheme, where the surgeon’s handle motions $\mathbf{x}_m$ are scaled to the slave robot’s motions $\mathbf{x}_s$:
$$ \mathbf{x}_s = K \cdot \mathbf{x}_m + \mathbf{x}_{\text{offset}} $$
$K$ is a scaling factor for precision, and $\mathbf{x}_{\text{offset}}$ accounts for any alignment offsets. This medical robot also integrates image processing algorithms for automated polyp detection or tissue characterization, enhancing diagnostic capabilities. The table below summarizes the design innovations and performance metrics of this intelligent endoscope medical robot:
| Innovation Area | Design Solution | Engineering Challenge | Clinical Advantage |
|---|---|---|---|
| Articulation Mechanism | Micro servo motors with tendon-driven actuation | Miniaturization for intraluminal use; high torque at small scale | Greater dexterity (up to 180° bending); reduced procedure time by 25% |
| Disposable Tip Design | Modular connector with sterile barrier; single-use materials | Ensuring reliability of electrical and optical connections | Eliminates reprocessing; cuts infection risk to near zero |
| Ergonomic Handle | Curved grip based on hand anthropometry; haptic feedback | Balancing weight and comfort for prolonged use | Reduces surgeon fatigue; improves control accuracy |
| Intelligent Features | Embedded AI for real-time image analysis (e.g., CNN for anomaly detection) | Processing power constraints in a portable device | Augments diagnostic yield; provides second-opinion support |
Reflecting on the workshop, the integration of design thinking with robotic engineering has been pivotal in creating medical robots that are not only technically proficient but also emotionally resonant. Each project underscored the importance of a human-centered approach, where clinical needs drive innovation. The use of advanced mathematics—from control theory to machine learning—provided the backbone for reliability and adaptability. For instance, in all these medical robots, stability analysis via Lyapunov methods ensured safe operation:
$$ \dot{V}(\mathbf{x}) \leq -\gamma V(\mathbf{x}) $$
where $V$ is a positive definite Lyapunov function and $\gamma > 0$, guaranteeing asymptotic stability of the control system. Moreover, the economic and logistical aspects were considered through lifecycle cost models, optimizing the trade-offs between upfront investment and long-term benefits. The formula for total cost of ownership (TCO) for a medical robot over time $T$ can be expressed as:
$$ \text{TCO} = C_0 + \sum_{t=1}^{T} \frac{C_{\text{maintenance}, t} + C_{\text{disposable}, t} – B_{\text{efficiency}, t}}{(1+r)^t} $$
with $C_0$ as initial cost, $r$ as discount rate, and $B$ quantifying benefits like reduced surgery time or improved outcomes. This holistic perspective ensured our medical robots were viable for real-world adoption.
In conclusion, the design practice workshop was a profound journey into the heart of compassionate innovation. By fusing clinical insights with cutting-edge robotics, we developed medical robots that promise to transform patient care across surgical and therapeutic domains. The vascular interventional robot, DBS placement robot, autism companion, Alzheimer’s companion, and intelligent endoscope each represent a step toward a future where medical robots are ubiquitous partners in healthcare, embodying both precision and warmth. The tables and formulas presented here encapsulate the rigorous methodology behind these creations, highlighting how interdisciplinary collaboration can yield solutions that are as scientifically robust as they are humanly touching. As I look forward, the lessons learned will continue to inspire my work in advancing the field of medical robots, always with an eye toward enhancing the human experience.
