The evolution of medical robot systems represents a pivotal convergence of engineering innovation and clinical necessity. As a cornerstone of high-end medical equipment, the integration of medical robot platforms with frontier technologies such as artificial intelligence, brain-computer interfaces, and advanced communications is fundamentally reshaping healthcare delivery. From my perspective, a comprehensive understanding of the current state and core technological underpinnings of medical robot development is crucial for strategically advancing the field, enhancing industrial competitiveness, and ultimately improving patient outcomes on a global scale. This article synthesizes a multidimensional analysis, examining the application landscape, dissecting the core technological pillars, and forecasting the critical breakthrough directions that will define the next generation of intelligent, accessible, and integrated medical robot ecosystems.
Current Application Landscape of Medical Robots
The application of medical robot systems is diversifying rapidly across the clinical spectrum. I categorize them primarily into four domains, each addressing distinct clinical needs and presenting unique engineering challenges. The following table summarizes their primary functions, advantages, and representative focus areas.
| Category | Primary Function | Key Advantages | Representative Applications |
|---|---|---|---|
| Surgical Robots | Assist in precise, minimally invasive interventions. | Enhanced precision, stability, tremor filtering, remote operation capability. | Laparoscopic, orthopedic, vascular, natural orifice, percutaneous surgery. |
| Rehabilitation Robots | Facilitate recovery of motor or cognitive function. | High-intensity, repetitive, measurable therapy; reduces therapist burden. | Upper/lower limb training, gait therapy, hand/wrist rehabilitation. |
| Diagnostic Robots | Aid in health monitoring, data acquisition, and disease identification. | Objective data analysis, access to confined spaces, integration with AI for pattern recognition. | Capsule endoscopy, AI-assisted medical image analysis, physiological monitoring. |
| Other Medical Service Robots | Perform auxiliary, non-therapeutic tasks in clinical settings. | Increases efficiency, reduces human exposure to hazards, ensures consistent task execution. | Disinfection, logistics/transport, patient assistance and feeding. |
Surgical Robots: Extending the Surgeon’s Capabilities
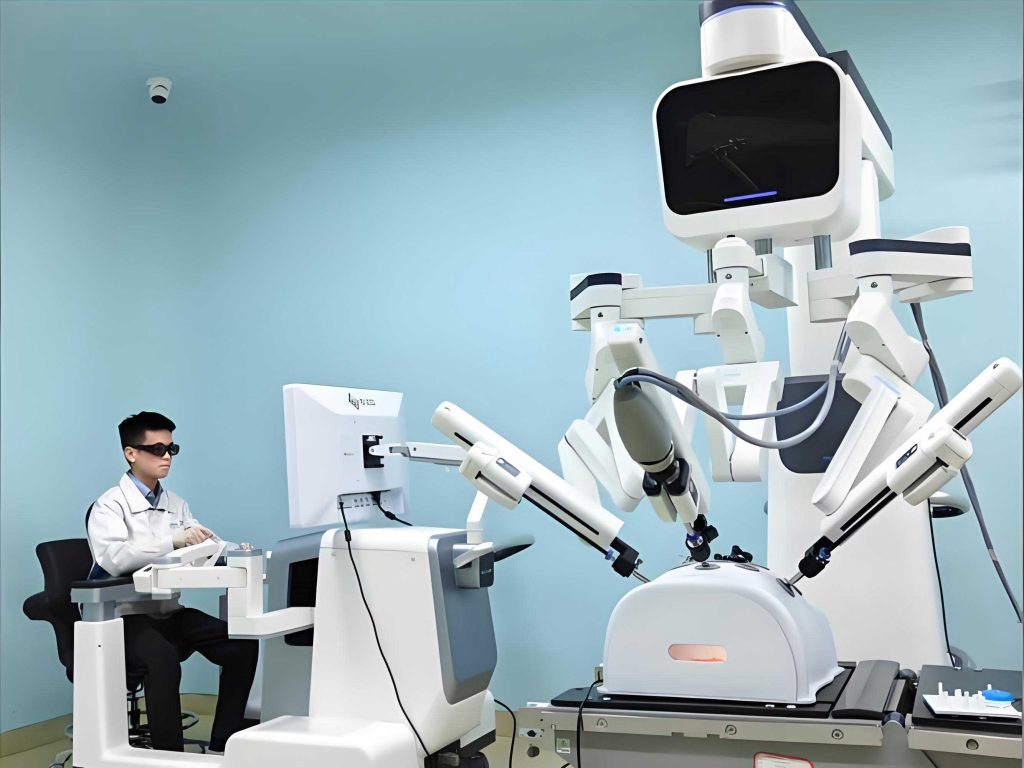
Surgical robots are perhaps the most prominent class, designed to augment a surgeon’s skills through enhanced dexterity, precision, and visualization. The core value proposition of a medical robot in surgery lies in translating the surgeon’s hand movements into scaled, filtered, and steady motions of miniature instruments inside the patient’s body.
- Laparoscopic Robotic Systems: These multi- or single-port systems are master-slave teleoperated platforms. The surgeon controls robotic arms equipped with high-dexterity wristed instruments from a console, viewing a magnified 3D high-definition field. Their design overcomes the limitations of traditional straight-stick laparoscopy, enabling complex dissection and suturing in confined spaces like the pelvis or chest.
- Orthopedic Surgical Robots: This category focuses on precision in bone-bound procedures. They typically integrate pre-operative planning software based on CT scans, intra-operative navigation (often using optical tracking), and a robotic arm to guide bone resection or implant placement. The fundamental kinematic model for tool positioning relative to the bone can be described as finding the transformation $T_{tool}^{bone}$ that satisfies the pre-operative plan:
$$ T_{tool}^{bone} = (T_{tracker}^{bone})^{-1} \cdot T_{tracker}^{camera} \cdot T_{ref}^{camera} \cdot T_{tool}^{ref} $$
where $T_{a}^{b}$ represents the transformation from frame *a* to frame *b*, involving the bone, optical tracker, camera, and tool reference frames.
- Interventional & Other Surgical Robots: This broad group includes systems for vascular procedures (guiding catheters and wires), bronchoscopy (navigating lung airways), and percutaneous needle placement (for biopsy or ablation). The common thread is the use of a medical robot to remotely or semi-autonomously navigate flexible or rigid tools through complex anatomical pathways under real-time imaging guidance, reducing physician exposure to radiation and improving targeting accuracy.
Rehabilitation Robots: Bridging Neuroscience and Engineering
Rehabilitation medical robot systems are grounded in theories of neuroplasticity and motor learning. Their objective is to provide controlled, intensive, and engaging therapy to promote functional recovery. I observe two main archetypes: end-effector-based robots, which guide a patient’s limb via a handle or footplate, and exoskeleton robots, which attach to and move in parallel with the patient’s limb segments. The dynamics of an exoskeleton interacting with a human limb can be modeled as:
$$ M(q)\ddot{q} + C(q, \dot{q})\dot{q} + G(q) + \tau_{dis} = \tau_{motor} + \tau_{human} $$
where $q$ represents the joint angles, $M$ is the inertia matrix, $C$ captures Coriolis and centrifugal forces, $G$ is gravity, $\tau_{dis}$ denotes disturbances, $\tau_{motor}$ is the torque applied by the medical robot, and $\tau_{human}$ is the interaction torque from the patient. The challenge is to design controllers that ensure safety and promote active participation, often through adaptive impedance or assist-as-needed strategies.
Diagnostic and Service Robots: Expanding the Scope of Automation
Beyond direct intervention, medical robot technology is automating critical support functions. Diagnostic robots, like ingestible capsule endoscopes, provide unprecedented access for visualization. AI-integrated diagnostic systems act as powerful counterparts, analyzing vast datasets from medical images or physiological signals to identify patterns imperceptible to the human eye. Furthermore, service robots are becoming indispensable in clinical environments. Disinfection robots using UV-C light or plasma, automated guided vehicles for logistics, and assistive robots for patient lifting are enhancing operational efficiency, reducing nosocomial infection risks, and alleviating the physical strain on healthcare workers.
Core Technological Pillars of Medical Robots
The sophisticated functionality of any medical robot rests upon the integration of several interdependent technological pillars. From my analysis, progress in these core areas directly translates to enhanced clinical capability and safety.
| Technology Pillar | Core Objective | Key Challenges | Enabling Methods & Components |
|---|---|---|---|
| Structural Design | Create mechanisms that are safe, dexterous, and adaptable to anatomical constraints. | Miniaturization, achieving high force/stiffness in flexible systems, ensuring inherent safety. | Continuum/soft robotics, smart materials (e.g., shape memory alloys), compliant mechanisms, lockable structures. |
| Motion & Force Control | Execute precise, stable, and compliant movements in dynamic interaction with tissue. | Modeling tissue mechanics, compensating for dynamic effects, implementing transparent teleoperation. | Impedance/Admittance control, force-position hybrid control, adaptive and robust control algorithms. |
| Perception & Sensing | Acquire multi-modal data (position, force, vision, physiological) from the robot, patient, and environment. | Miniaturizing sensors, achieving biocompatibility, multi-modal sensor fusion, providing realistic haptic feedback. | Fiber Bragg Grating (FBG) sensors, flexible electronic skins, EM/optical trackers, advanced imaging (OCT, micro-US). |
| Information Processing & Navigation | Fuse data to create actionable models, plan optimal paths, and guide tools in real-time. | Real-time image registration, dealing with tissue deformation, uncertainty in perception. | AI/ML for image segmentation, SLAM in deformable environments, Kalman/particle filters, augmented reality overlay. |
| Human-Robot Interaction (HRI) & Teleoperation | Create intuitive, low-latency interfaces for natural control and effective situational awareness. | Mitigating communication latency, designing intuitive control mappings, preventing cyber-security breaches. | Predictive control displays, haptic feedback devices, gaze-tracking, 5G/edge computing, shared control paradigms. |
| Artificial Intelligence | Embed cognitive functions for decision support, autonomy, and personalized adaptation. | Limited & biased datasets, “black-box” models, integration into clinical workflow, regulatory approval. | Deep learning for vision, reinforcement learning for control, natural language processing, explainable AI (XAI). |
Deep Dive: Motion Control and Haptics
The control system is the “central nervous system” of a medical robot. In teleoperated systems, the goal is to achieve transparency: making the surgeon feel as if they are directly manipulating the tissue. This requires both accurate motion tracking and faithful force feedback. A standard bilateral teleoperation control law aims to satisfy the following ideal relationships:
$$ x_s = k_x x_m, \quad F_m = k_f F_s $$
where $x_m$ and $x_s$ are master and slave positions, $F_m$ and $F_s$ are forces on the master and slave, and $k_x$, $k_f$ are scaling factors. In reality, dynamics, communication delays ($T_d$), and stability concerns complicate this. The passivity-based Time-Domain Passivity Control (TDPC) approach is often used to guarantee stability despite delay:
$$ E_{in}(t) – E_{out}(t) = \int_{0}^{t} (F_m(\tau)\dot{x}_m(\tau) – F_s(\tau-T_d)\dot{x}_s(\tau-T_d)) d\tau \geq 0 $$
This ensures the network energetically does not generate energy, preventing oscillations. Furthermore, for autonomous functions like maintaining a steady trajectory or applying a specific force profile, hybrid force/position control is essential. The controller often switches or blends between a position-controlled subspace and a force-controlled subspace defined by a selection matrix $S$:
$$ \tau = J^T ( S F_{desired} + (I-S) K_p (x_{desired} – x) ) $$
where $J$ is the Jacobian, $K_p$ is a position gain, and $S$ is a diagonal matrix with 1s for force-controlled degrees of freedom and 0s for position-controlled ones.
Deep Dive: The Role of AI and Navigation
Artificial intelligence is transforming every pillar, most visibly in perception and navigation. For a medical robot navigating inside the body, real-time “sense-plan-act” cycles are critical. AI enables semantic understanding of the scene. For instance, in bronchoscopy, a deep learning model can segment the airway tree from a CT scan and identify the target nodule. During the procedure, a visual SLAM (Simultaneous Localization and Mapping) algorithm, often based on convolutional neural networks (CNNs) or transformers, can estimate the endoscope’s pose within the deforming airways by comparing real-time video to the pre-operative model:
$$ \hat{T}_t = \underset{T}{\mathrm{argmin}} \sum_i \left\| I_{live}(\pi(T \cdot P_i)) – I_{CT}(P_i) \right\|^2 $$
Here, $\hat{T}_t$ is the estimated pose at time $t$, $P_i$ are 3D points from the CT model, $\pi$ is the projection function, and $I_{live}$ and $I_{CT}$ are the live and CT-derived virtual images. Reinforcement Learning (RL) is also emerging for autonomous control tasks, where the medical robot learns a policy $\pi_{\theta}(a|s)$ that maps states $s$ (e.g., image, position) to actions $a$ (e.g., joint velocities) to maximize a reward $r$ (e.g., reaching target without collision):
$$ J(\theta) = \mathbb{E}_{\tau \sim \pi_{\theta}} \left[ \sum_{t=0}^{T} \gamma^t r(s_t, a_t) \right] $$
The policy parameters $\theta$ are optimized to maximize the expected cumulative reward $J(\theta)$. While full autonomy is distant, AI-driven assistance for tool tracking, tissue classification, and warning generation is already enhancing the capabilities of the modern medical robot.
Critical Future Breakthrough Directions
Based on the trajectory of current research and persistent clinical needs, I identify four interconnected vectors that will drive the next wave of innovation in medical robot technology.
1. Intelligence and Autonomy: From Tool to Collaborative Partner
The future medical robot will evolve from a precise but passive tool into an intelligent system with situational awareness and decision-support autonomy. This involves moving beyond pre-programmed motions to context-aware operation. Key requirements include:
- Advanced Perception: Fusing multi-modal data (vision, touch, ultrasound, physiology) to create a real-time, holistic “clinical scene understanding.”
- Reasoning and Planning: Using symbolic AI and causal models alongside deep learning to reason about surgical steps, predict tissue behavior, and generate safe motion plans that adapt to intra-operative findings.
- Shared Autonomy: Developing fluid control paradigms where responsibility is dynamically traded between the surgeon and the medical robot based on task complexity, surgeon intent, and patient state. The robot might autonomously perform a repetitive stabilization task while the surgeon focuses on a critical dissection.
The mathematical formulation could involve a partially observable Markov decision process (POMDP) where the robot maintains a belief state $b(s)$ over the true world state and chooses actions to maximize a reward function that encodes clinical goals and safety constraints.
2. Miniaturization and Softness: The Era of Embodied and Bio-Inspired Design
Accessing deeper and more delicate regions of the body necessitates a fundamental shift in medical robot morphology. The drive is towards milli-/micro-scale robots and soft, compliant structures.
- Micro/Nanorobots: These are untethered agents designed for targeted drug delivery, sensing, or micro-manipulation. Propulsion and control often use external fields (magnetic, acoustic). The magnetic force on a spherical microrobot is given by: $$ F_m = (m \cdot \nabla) B $$ where $m$ is the magnetic moment and $B$ is the external magnetic field. Precise swarm control and imaging remain significant hurdles.
- Soft Robotics: Using compliant materials, pneumatic actuators, and tendon-driven mechanisms allows a medical robot to conform safely to anatomy. The challenge is modeling and controlling these highly nonlinear systems. The mechanics are often described using Cosserat rod theory or finite element methods. Variable stiffness mechanisms, often using jamming of granular media or low-melting-point alloys, are crucial to combine safe interaction with the ability to apply necessary forces.
3. Multimodal Interaction and Ubiquitous Telepresence
The interface between human and medical robot will become richer and more immersive, while geographical barriers will further dissolve.
- Multimodal HRI: Control will integrate not just hands, but also gaze, voice, and even decoded neural signals. Feedback will extend beyond visual and force cues to include tactile textures and auditory cues. The integration follows a sensor fusion framework: $$ \hat{y} = f( V, H, A, P; \Theta ) $$ where the estimated user intent $\hat{y}$ is a function $f$ (e.g., a neural network with parameters $\Theta$) of visual ($V$), haptic ($H$), auditory ($A$), and physiological ($P$) inputs.
- Robust Teleoperation: Next-generation networks (5G/6G, satellite) with ultra-reliable low-latency communication (URLLC) and edge computing will make remote surgery more feasible and safe. The focus will be on predictive simulation and “ghost robotics,” where a digital twin of the remote site predicts outcomes to compensate for latency, allowing the local surgeon to operate in apparent real-time.
4. System Integration and Ecosystem Synergy
The isolated medical robot will become a node in a broader, intelligent medical ecosystem. This involves:
- Interoperability and Standards: Developing common communication protocols (like ROS-2 in healthcare) and data formats to allow robots from different manufacturers to work with hospital information systems, imaging archives (PACS), and other robotic devices seamlessly.
- Digital Twin and Cognitive Orthopedics: Creating patient-specific digital twins that simulate procedures pre-operatively and update in real-time during surgery using sensor data. This allows for predictive analytics (“what-if” scenarios) and personalized surgical planning.
- Data-Centric Ecosystem: Federated learning will enable medical robot AI models to learn from vast, distributed datasets across hospitals without sharing raw patient data, addressing privacy concerns while improving algorithm robustness and generalizability. The learning objective across $N$ institutions becomes: $$ \min_{\theta} \sum_{i=1}^{N} \frac{n_i}{n} F_i(\theta) $$ where $F_i(\theta)$ is the local loss function at institution $i$ with $n_i$ samples, $n$ is the total samples, and $\theta$ are the global model parameters updated via secure aggregation.
Conclusion
In my view, the field of medical robot technology stands at an inflection point, propelled by deep interdisciplinary convergence. The journey from mechanically assisted tools to intelligent, networked partners in care is well underway. The core technological pillars—spanning innovative mechanical design, advanced control, rich perception, AI-driven navigation, and intuitive interaction—are experiencing rapid, synergistic advancement. The future trajectory, focused on embodied intelligence, miniaturized and soft form factors, seamless multimodal telepresence, and integrated system ecosystems, promises to fundamentally reshape surgical practice, rehabilitative therapy, diagnostic pathways, and hospital operations. Realizing this future will require sustained collaboration across engineering, computer science, clinical medicine, and regulatory bodies. By strategically focusing on these breakthrough directions, we can accelerate the development of a new generation of medical robot systems that are not only more capable and accessible but also inherently safer and more responsive to the nuanced needs of individual patients, thereby solidifying their role as a transformative force in global healthcare.
