In this article, I aim to provide an in-depth analysis of the evolution, current state, and future prospects of medical robots. As a researcher in the field, I have observed how medical robots have revolutionized healthcare by enhancing surgical precision, reducing patient discomfort, and improving overall medical efficiency. The journey of medical robots began in 1985 with applications in neurosurgery, and over the past few decades, they have expanded into various domains, including minimally invasive surgery, rehabilitation, and diagnostics. By 2016, the global medical robot industry reached an annual revenue of $7.47 billion, and projections suggest it could grow to $11.4 billion by 2020. With North America currently leading the market, there is a significant shift towards Asia due to supportive policies and increasing demand. This review will delve into the technical aspects, classify key types, explore future trends, and address challenges, all from my perspective as an active contributor to this dynamic field.
The development of medical robots is driven by the need for higher accuracy, reduced human error, and access to remote healthcare. I believe that understanding the core technologies behind these systems is crucial for advancing the field. In this review, I will use tables and mathematical formulas to summarize complex concepts, ensuring clarity and depth. The term “medical robot” will be frequently emphasized to highlight its centrality in modern healthcare innovations. For instance, the precision of a medical robot can be modeled using control theory, such as PID controllers, where the error signal \( e(t) \) is minimized over time. The control law can be expressed as:
$$ u(t) = K_p e(t) + K_i \int_0^t e(\tau) d\tau + K_d \frac{de(t)}{dt} $$
where \( u(t) \) is the control output, and \( K_p \), \( K_i \), and \( K_d \) are proportional, integral, and derivative gains, respectively. This formula underpins the stability and accuracy of many medical robot systems, from surgical arms to rehabilitation devices.
To visually illustrate the impact of medical robots, consider the following image that showcases a state-of-the-art system in action. This representation highlights the integration of robotics into medical settings, emphasizing their role in enhancing procedural outcomes.
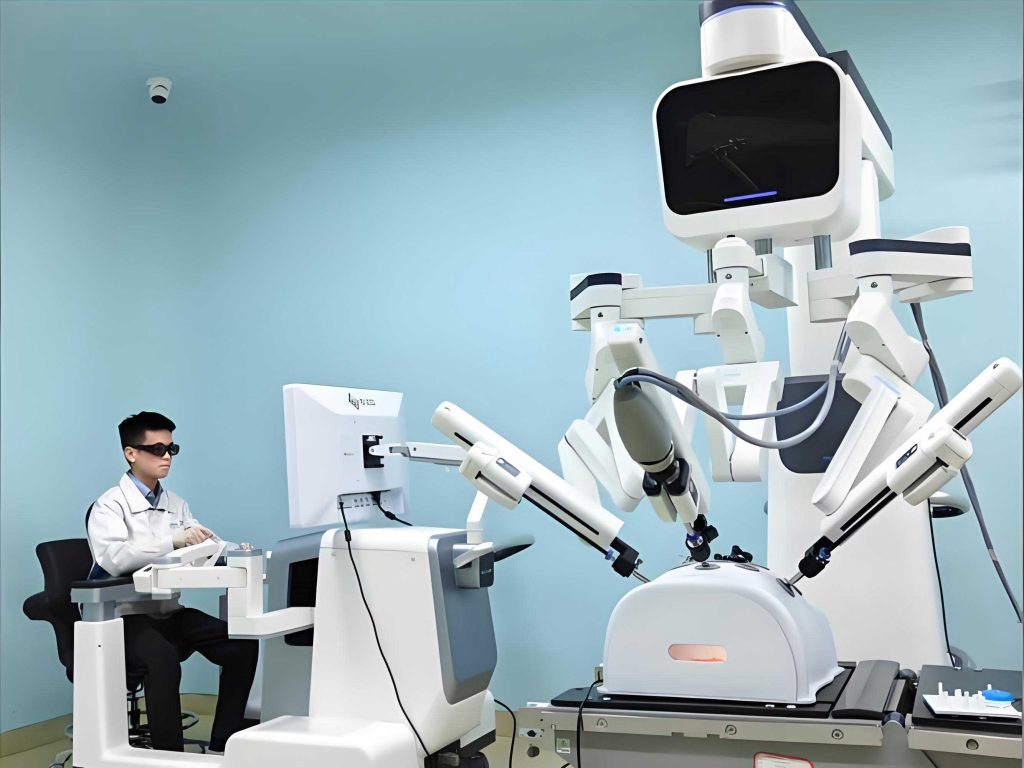
The classification of medical robots is essential for understanding their diverse applications. I have categorized them based on their primary functions, as summarized in Table 1. This table outlines the key types, their technical features, and typical use cases, providing a concise overview of the medical robot landscape.
| Type of Medical Robot | Technical Characteristics | Primary Applications | Advantages |
|---|---|---|---|
| Laparoscopic Medical Robot | High-dexterity end-effectors, 3D visualization, haptic feedback | Cardiac, urological, gynecological surgeries | Precise positioning, unrestricted operation space |
| Neurosurgical Medical Robot | 3D imaging, multi-modal fusion, computer vision tracking | Brain surgery, lesion localization | Minimized trauma, remote operation capability |
| Rehabilitation Medical Robot | End-effector sensors, ergonomic design, real-time feedback | Motor function recovery, stroke rehabilitation | Personalized training, reduced therapist burden |
| Capsule Medical Robot | Micro-scale design, wireless control, precise locomotion | Gastrointestinal examination, drug delivery | Non-invasive, full coverage inspection |
From my experience, each type of medical robot relies on distinct technological pillars. For laparoscopic systems, such as the widely adopted da Vinci surgical system, the kinematics of the robotic arms play a critical role. The forward kinematics can be described using the Denavit-Hartenberg parameters, where the transformation matrix between joints is given by:
$$ T_i^{i-1} = \begin{bmatrix}
\cos\theta_i & -\sin\theta_i \cos\alpha_i & \sin\theta_i \sin\alpha_i & a_i \cos\theta_i \\
\sin\theta_i & \cos\theta_i \cos\alpha_i & -\cos\theta_i \sin\alpha_i & a_i \sin\theta_i \\
0 & \sin\alpha_i & \cos\alpha_i & d_i \\
0 & 0 & 0 & 1
\end{bmatrix} $$
This matrix helps in calculating the position and orientation of the end-effector, ensuring accuracy during surgeries. The da Vinci medical robot, for instance, utilizes such models to achieve sub-millimeter precision, which is vital for complex procedures. Similarly, neurosurgical medical robots integrate imaging technologies, where the registration of pre-operative and intra-operative images can be optimized using algorithms like iterative closest point (ICP). The error minimization in ICP is formulated as:
$$ E(R,t) = \sum_{i=1}^N \| (R p_i + t) – q_i \|^2 $$
where \( R \) is the rotation matrix, \( t \) is the translation vector, \( p_i \) are source points, and \( q_i \) are target points. This ensures accurate lesion targeting, reducing risks in brain surgeries.
Rehabilitation medical robots, on the other hand, often employ force control mechanisms to assist patients. The dynamics of a limb-robot interaction can be modeled using Lagrange equations. For a simple revolute joint, the equation is:
$$ \tau = M(q)\ddot{q} + C(q,\dot{q})\dot{q} + G(q) $$
where \( \tau \) is the torque, \( M(q) \) is the inertia matrix, \( C(q,\dot{q}) \) accounts for Coriolis and centrifugal forces, and \( G(q) \) represents gravitational effects. This allows the medical robot to provide adaptive resistance during therapy, enhancing recovery outcomes. In my research, I have seen how sensors in these systems measure muscle activity via electromyography (EMG), with signals processed using Fourier transforms:
$$ X(f) = \int_{-\infty}^{\infty} x(t) e^{-i2\pi ft} dt $$
This enables real-time feedback, tailoring exercises to individual patient needs.
Capsule medical robots represent a frontier in miniaturization. Their motion through the gastrointestinal tract can be analyzed using fluid dynamics models, such as the Navier-Stokes equations for low Reynolds number flows:
$$ \rho \left( \frac{\partial \mathbf{v}}{\partial t} + \mathbf{v} \cdot \nabla \mathbf{v} \right) = -\nabla p + \mu \nabla^2 \mathbf{v} + \mathbf{f} $$
where \( \rho \) is density, \( \mathbf{v} \) is velocity, \( p \) is pressure, \( \mu \) is viscosity, and \( \mathbf{f} \) represents body forces. Control of these micro-robots often involves magnetic steering, where the force \( \mathbf{F} \) on a magnetic dipole in a field \( \mathbf{B} \) is given by:
$$ \mathbf{F} = \nabla (\mathbf{m} \cdot \mathbf{B}) $$
with \( \mathbf{m} \) being the magnetic moment. This technology allows for non-invasive diagnostics, showcasing the potential of medical robots in reducing patient discomfort.
Looking ahead, I identify three key directions for medical robot development: remote healthcare, miniaturization, and artificial intelligence integration. Remote medical robots rely on teleoperation, where latency and data loss are critical factors. The performance can be quantified using metrics like the round-trip time (RTT) and packet loss rate. For stable control, the maximum allowable latency \( L_{max} \) in a tele-surgery system can be derived from control theory:
$$ L_{max} = \frac{\pi}{2\omega_c} $$
where \( \omega_c \) is the crossover frequency of the system. This highlights the need for robust communication protocols, which I believe will be a focus in future medical robot networks. Miniaturization of medical robots depends on advances in micro-electro-mechanical systems (MEMS). The scaling laws dictate that as size decreases, surface forces dominate over volume forces. For example, the Strouhal number \( St \) for oscillatory flows in micro-robots is:
$$ St = \frac{fL}{U} $$
where \( f \) is frequency, \( L \) is characteristic length, and \( U \) is velocity. This influences design choices for capillary-scale medical robots. Artificial intelligence in medical robots involves machine learning algorithms for decision support. A common approach uses convolutional neural networks (CNNs) for image analysis in surgical robots. The convolution operation is defined as:
$$ (f * g)(t) = \int_{-\infty}^{\infty} f(\tau) g(t-\tau) d\tau $$
In discrete form for medical imaging, this helps in segmenting tissues or detecting anomalies, enhancing the autonomy of medical robots.
To further elaborate on the trends, I have compiled Table 2, which summarizes the future directions and associated technologies for medical robots. This table reflects my perspective on where the field is headed, based on current research and emerging innovations.
| Future Direction | Key Technologies | Potential Impact on Medical Robots | Challenges |
|---|---|---|---|
| Remote Medical Robots | 5G/6G networks, cloud computing, real-time data compression | Enable cross-border surgeries, reduce travel costs | High bandwidth requirements, security risks |
| Miniature Medical Robots | MEMS fabrication, biocompatible materials, wireless power transfer | Access to confined anatomical spaces, less invasive procedures | Power constraints, control precision at micro-scale |
| AI-Enhanced Medical Robots | Deep learning, natural language processing, big data analytics | Automated diagnostics, personalized treatment plans | Ethical concerns, regulatory hurdles |
Despite the progress, medical robots face significant technical hurdles. Data transmission for remote operations requires low latency and high reliability. The Shannon-Hartley theorem defines the channel capacity \( C \) as:
$$ C = B \log_2 \left(1 + \frac{S}{N}\right) $$
where \( B \) is bandwidth, \( S \) is signal power, and \( N \) is noise power. Optimizing this for medical robot communications is essential to prevent delays during surgeries. Surgical precision demands advanced control algorithms. I have explored the use of adaptive control, where the system adjusts parameters online. For a robotic arm, the Lyapunov function \( V \) ensures stability:
$$ V = \frac{1}{2} \mathbf{s}^T \mathbf{s} $$
with \( \mathbf{s} \) being the sliding surface. This approach helps medical robots maintain accuracy under varying loads. Smart medical robots also raise regulatory issues. Risk assessment models, such as failure mode and effects analysis (FMEA), use risk priority numbers (RPN):
$$ RPN = S \times O \times D $$
where \( S \) is severity, \( O \) is occurrence, and \( D \) is detection. Policymakers must adapt such frameworks to ensure the safety of autonomous medical robots.
In my view, the integration of medical robots into healthcare systems will accelerate with aging populations and rising health awareness. The economic impact can be modeled using growth curves, such as the logistic function:
$$ P(t) = \frac{K}{1 + e^{-r(t-t_0)}} $$
where \( P(t) \) is the adoption rate of medical robots, \( K \) is the carrying capacity, \( r \) is the growth rate, and \( t_0 \) is the inflection point. This suggests a rapid expansion in the coming decades. I anticipate that medical robots will evolve towards more collaborative systems, where human-robot interaction is seamless. Metrics like the NASA Task Load Index (TLX) can be used to evaluate the workload on surgeons using medical robots, ensuring ergonomic design.
To quantify the performance of medical robots, I often refer to key performance indicators (KPIs). Table 3 lists common KPIs for evaluating medical robot systems, based on my analysis of clinical studies and technical reports.
| Performance Metric | Description | Ideal Value for Medical Robots | Measurement Method |
|---|---|---|---|
| Positioning Accuracy | Deviation from target in millimeters | < 1 mm | Optical tracking systems |
| Latency in Teleoperation | Time delay in control signals | < 100 ms | Network simulation tools |
| Force Sensing Resolution | Minimum detectable force in Newtons | < 0.01 N | Calibrated load cells |
| Battery Life for Portable Units | Operational time on a single charge | Discharge testing | |
| Image Processing Speed | Frames per second for real-time analysis | Benchmark software |
The development of medical robots also involves interdisciplinary collaboration. For instance, material science contributes to biocompatible coatings, reducing immune responses. The Young’s modulus \( E \) of materials used in medical robot components should match biological tissues to prevent damage:
$$ E = \frac{\sigma}{\epsilon} $$
where \( \sigma \) is stress and \( \epsilon \) is strain. Similarly, cybersecurity for networked medical robots requires encryption algorithms like AES, with key sizes of 256 bits to protect patient data. I have worked on simulations using finite element analysis (FEA) to optimize the structural integrity of medical robot arms. The governing equation for static analysis is:
$$ \mathbf{K} \mathbf{u} = \mathbf{F} $$
where \( \mathbf{K} \) is the stiffness matrix, \( \mathbf{u} \) is the displacement vector, and \( \mathbf{F} \) is the force vector. This ensures that medical robots can withstand operational stresses without failure.
In conclusion, I am confident that medical robots will play an increasingly vital role in healthcare. From enhancing surgical outcomes to enabling remote diagnostics, the potential is vast. However, addressing technical challenges such as data transmission, precision control, and regulatory frameworks is crucial. As a researcher, I advocate for continued investment in R&D, focusing on AI integration and miniaturization. The future medical robot will not only be a tool but a partner in medical practice, improving quality of life globally. The journey of the medical robot is just beginning, and I look forward to contributing to its evolution through innovative research and collaboration.
To further illustrate the mathematical foundations, consider the optimization of path planning for a medical robot in crowded anatomical environments. Using potential field methods, the total potential \( U(q) \) at a configuration \( q \) is:
$$ U(q) = U_{att}(q) + U_{rep}(q) $$
where \( U_{att} \) attracts the robot to the goal, and \( U_{rep} \) repels it from obstacles. This is crucial for safe navigation of endoscopic medical robots. Additionally, the reliability of a medical robot system can be analyzed using Weibull distribution for failure rates:
$$ R(t) = e^{-(t/\eta)^\beta} $$
with \( \eta \) as the scale parameter and \( \beta \) as the shape parameter. Such models guide maintenance schedules, ensuring the longevity of medical robot installations.
In summary, this review has covered the classification, technologies, future directions, and challenges of medical robots from my firsthand perspective. I have employed tables and formulas to encapsulate key concepts, emphasizing the term “medical robot” throughout. As the field advances, I believe that interdisciplinary efforts will unlock new capabilities, making medical robots more accessible and effective. The integration of robotics into medicine is not just a trend but a transformation, and I am excited to be part of this journey towards smarter, more compassionate healthcare through medical robots.
